 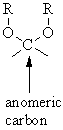
If it is directed in the opposite direction it is the β-anomer. In a Fischer projection, if the substituent off the anomeric centre is on the same side as the oxygen of the configurational (D- or L-) carbon, then it is the α-anomer. The configuration at the anomeric centre (that derived from the carbonyl carbon) is denoted alpha- (α-) or beta- (β-) by reference to the stereocentre that determines the absolute configuration. They are diastereoisomers of one another. The two stereoisomers formed from the two possible stereochemistries at the anomeric centre are called anomers. The anomeric centre of a sugar is a stereocentre created from the intramolecular formation of an acetal (or ketal) of a sugar hydroxyl group and an aldehyde (or ketone) group. If you would like to suggest an addition or correction, please contact the page's Responsible Curator directly by e-mail. CAZypedia is a living document, so further improvement of this page is still possible. Henry Rzepa., "Anomeric effects at boron, silicon and phosphorus.", 2016.This page has been approved by the Responsible Curator as essentially complete. So this little exploration shows that the anomeric effect, best known for sugars and at a carbon centre, is in fact more general to the adjacent elements. As with Si, the ridge in the 60° region run orthogonal to the diagonal. The two off-diagonal clusters show just one such orientation. This shows four clusters the two on the diagonal show respectively two and no antiperiplanar interactions. Most of the hits are in fact for P there are none for N. : a cyclic stereoisomer of a carbohydrate with isomerism involving only the arrangement of atoms or groups at the aldehyde or ketone position anomeric an-mer-ik adjective Dictionary Entries Near anomer anomaly anomer anomia See More Nearby Entries Cite this Entry Style Anomer. 
If its present, the effect is small.įinally 4-coordinate group 15 elements. Quora Answer (1 of 2): As far as I know, anomeric carbon is used to refer only to cyclized forms of sugars. Did you actually mean anorexic or anorexia Wiktionary Rate this definition: 0.0 / 0 votes anomeric adjective Of, or pertaining to anomers. This is not a carbohydrate because the molecule does not contain an aldehyde or a ketone functional group. Definitions for anomeric anomer·ic This dictionary definitions page includes all the possible meanings, example usage and translations of the word anomeric. Solution This is a carbohydrate because the molecule contains an aldehyde functional group with OH groups on the other two carbon atoms. For ease of viewing, the six-membered hemiacetal structure is drawn as a flat hexagon, but it actually assumes a chair conformation. Since the off-diagonal clusters are relatively prominent, implying just one anomeric interaction, it is of interest to see if this results in any asymmetry in the two Si-O bond lengths. Which compounds would be classified as carbohydrates 1. An interesting point to follow up perhaps? Notice also that whereas the torsions in the region of 60° for the carbon centre lie along a ridge coincident with the diagonal (bottom left to top right), that for the silicon centre show a ridge running orthogonal to the diagonal. In carbohydrate chemistry, a pair of anomers (from Greek 'up, above', and 'part') is a pair of near-identical stereoisomers or diastereomers that differ at only the anomeric carbon, the carbon that bears the aldehyde or ketone functional group in the sugar's open-chain form. So here, the anomeric effect is much stronger. The previous hotspot for which both measured torsions were 180° is largely absent. Again three significant clusters are seen one with two antiperiplanar lone pair alignments with Si-O bonds, and two more with just one such alignment. The next centre is at 4-coordinate silicon. This compares to the carbon-anomeric plot which is shown here for comparison, where the top right cluster of 180° torsions contains proportionately few hits than with boron. So yes, 4-coordinate boron can exhibit an anomeric effect! There are two more diffuse clusters where only one antiperiplanar alignment is seen. This latter is the one that implies that there must be two lone pairs, one on each oxygen, that are anti-periplanar to the adjacent B-O bond. The result shows two prominent clusters, one with both torsion angles being 180°, and another with both being ~60°. I start with 4-coordinate boron, using the crystal structure search definition below (along with R < 0.1, no disorder, no errors). 
Here I explore whether other centres can exhibit the phenomenon. If it is down (axial), the sugar is in the form, while if it is up (equatorial). The anomeric effect occurs at 4-coordinate (sp 3) carbon centres carrying two oxygen substituents and involves an alignment of a lone electron pair on one oxygen with the adjacent C-O σ*-bond of the other oxygen. Also the hydroxyl substituent of the anomeric carbon can be either up or down. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
